Scientists can’t technically create a new color, but they can create new materials that will refract and absorb light in new ways. In 2009, Oregon State chemist Dr. Mas Subramanian and his research team discovered a means of producing a brand new shade of blue. It was the first to be synthesized in a lab in over 200 years. The last blue discovery was cobalt blue, created by French chemist Louis Jacques Thenard in 1802.
Subramanian’s brand new blue was dubbed YInMn Blue. The name comes from yttrium, indium, and manganese, which are the elements that make up the pigment. The team at Oregon State discovered this completely by accident. They’d been researching the electrical properties of manganese oxides.
“We made a series of compounds by combining oxides of three elements: yttrium (white), indium (yellow) and manganese (black), and heating the mixtures to very high temperatures of 1250 Celsius, or 2300 Fahrenheit, to study their magnetic properties,” explains Subramanian. The results of these experiments were not what they’d expected. “The student pulled the samples from the furnace and they came out as stunningly intense blue powders. Imagine my surprise—because we had started with materials that were white, yellow, and black in color.”
Knowing that simply mixing two pigments together is not enough to produce a new color, and not having seen such a vivid blue in his 35 years of research, Subramanian immediately knew this was an important breakthrough.
The color blue has always been sought after. Due to the challenges of extracting blue color from natural materials, it is thought to be the first human-made synthetic pigment, and was first produced in ancient Egypt in the third millennium BC. Blue is still the color scientists have have the most difficulty synthesizing, and is notoriously unstable as an inorganic compound. But probably the most surprising thing about blue is that it doesn’t occur in the natural world.
The sky, the ocean, bluebells, the feathers of certain birds, and eyes all may certainly look blue, but as Subramanian explains, “There is no actual pigment in them.” Naturally occurring shades of blue are not blue in the same way that leaves are green or poppies are red. Their color comes from a trick of structure. Unlike various reds, browns, and yellows, you can’t grind blue up and put it on the canvas, which is why the team at Oregon State University knew they were onto something important with YInMn Blue.
YInMn Blue’s crystal formation makes it superior to other compositions of inorganic pigments. It is stable, resilient to acid and alkali, durable, heat and UV resistant, and non-toxic. Unlike other blues, it won’t fade over time. It’s this vibrancy that makes YInMn Blue especially attractive to artists and designers. It’s particularly useful in art restoration, as it is a similar tone to ultramarine blue, which is a pigment traditionally used in painting—but YInMn Blue will last much longer.

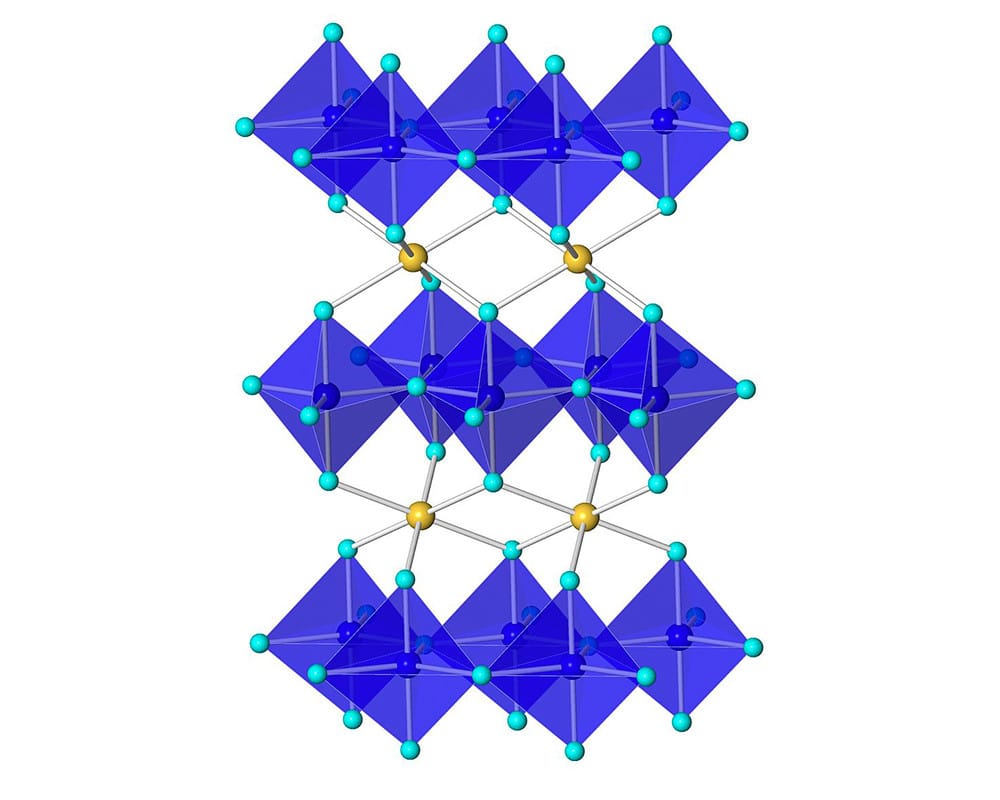
Via Oregon State’s chemistry department: “The chemical formula of YInMn Blue is YIn1-xMnxO3. These compositions adopt a crystal structure in which the chromophore responsible for the intense blue color (Mn3+) resides in the trigonal bipyramidal site (shown in blue).”
The color also has potential for some unique industrial uses. Subramanian explains that YInMn Blue’s ability to reflect heat “is a bonus, as it can reduce the cost of cooling buildings and cars.” Utilized in roofing, for example, this new blue could play an important part in energy efficiency and environmentally friendly heating technologies.
Taking this pigment out of the lab and onto the market still requires additional testing and tweaking, which is where The Shepherd Color Company comes into the picture. Specializing in high performance inorganic colour compounds, they have the ability, experience, and technology to produce the colour on a large scale. Shepherd is currently still testing YInMn blue for levels of toxicity and waiting for EPA approval before making the colour widely available.
Shepherd Color Company’s marketing manager Mark Ryan explains the latest developments in the industry: “We patented the NTP (Niobium Tin Pyrochlore) yellow pigments a few years ago to replace lead chromate. It really is an incredible invention because of its durability, chromaticity, and opacity.” Ryan also tells us that yellow isn’t the only color to be upgraded. “Due to European REACH chemical restrictions, we have bought out new green pigments that reduce exposure to lead and chrome compounds.”
To stay up to date with industry standards and regulations, research institutions and colour companies like TSCC and Pantone invest countless hours into researching and developing ways to improve existing compounds. The ideal pigment is one that is thermally stable, chemically inert, non-toxic, non-carcinogenic, and still vibrant. By applying the same strong and stable trigonal bi-pyramidal structure that YInMn blue uses to other compounds, the research team at Oregon State University have made some exciting progress in this area.
“We have made other colours,” says Subramanian. “By replacing iron for manganese we have made orange, by replacing manganese with copper we have made green and by swapping indium with titanium and zinc we have made purple.”
After blue, the search for a perfect red pigment is something of a Holy Grail in the color industry. “Currently we are looking for a non-toxic, stable red pigment. The inorganic red pigments used currently are not very stable, and frequently have toxic elements such as cadmium, lead, and mercury in them”.
Subramanian’s team is working to create a red oxide like YInMn Blue, with certain energy gaps between the carbon atoms. An ideal red would, like this new blue, be less toxic and also have potential to be used in the energy sector. Perhaps sometime in the near future we will be hearing about a new cadmium or vermilion red that helps heat homes more efficiently. In the meantime, we can wait for YInMn Blue to hit the commercial market. Crayola recently announced that it will be debuting a YInMn Blue crayon later this year.
More on color:
Stuart Semple is Winning the Art World Feud Over the Blackest Black
Photographs of the Ugliest Color in the World
Photos of Pantone’s Super Green 2017 Color of the Year












